Research: Amyloid biotechnology
Amyloid fibrils may not only occur inside living organismus in the course of disease, but also as functional devices. Likewise there are increasing attempts to utilize amyloids for technological purposes. We previously extended this research by analyzing the applicative potential of amyloid oligomers. Moreover, we have been using biotechnologically engineered antibody fragments to investigate the mechanism and pathogenicity of different amyloid structures.
Selected references:
Luong TQ, Erwin N, Neumann M, Schmidt A, Loos C, Schmidt V, Fändrich M, Winter R Hydrostatic pressure increases the catalytic activity of amyloid fibril enzymes.
Angewandte Chemie Int. Ed. 2016, 55, 12412–12416
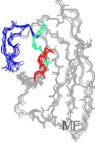
Morgado I, Wieligmann K, Bereza M, Rönicke R, Meinhardt K, Wacker J, Hortschansky P, Malešević M, Parthier C, Schiene-Fischer C, Reymann KG, Stubbs MT, Görlach M, Horn U, Fändrich M
Molecular Basis of β-Amyloid Oligomer Binding and Inhibition with a Conformation-Specific Antibody Fragment.
Proc. Natl. Acad. Sci. U.S.A. 2012, 109, 12503-12508
Contact

Prof. Dr. Marcus Fändrich
Director of the Institute
Tel: +49-(0)731/50 32750
Fax: +49-(0)731/50 32759
E-Mail: marcus.faendrich
Helmholtzstraße 8/1
Room number 1.55
