What it takes to cross borders
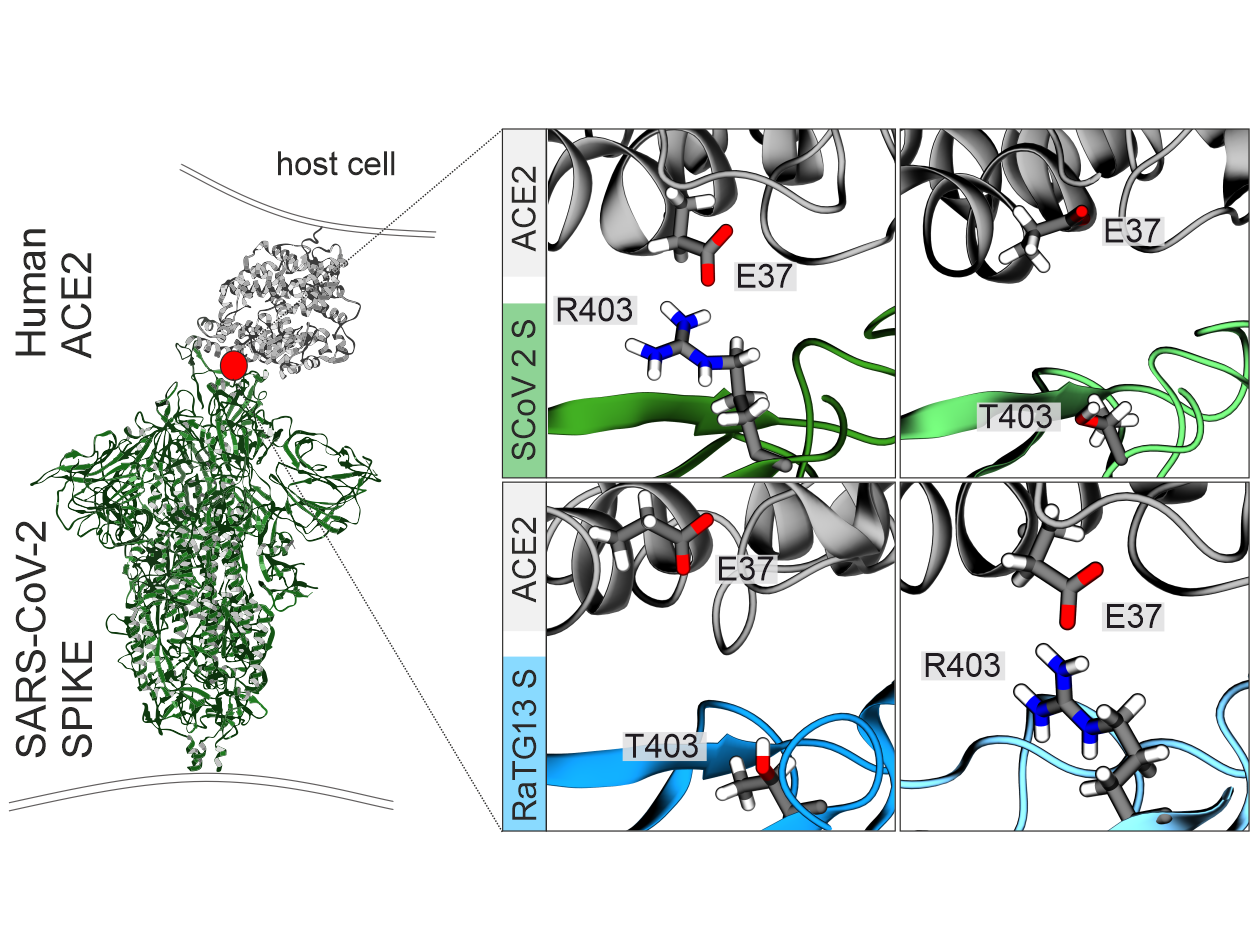
Bat coronaviruses have crossed the species barrier to humans on several occasions and are the origin of the COVID-19 pandemic. With over 96% sequence identity, the bat sarbecovirus RaTG13 is one of the closest relatives of SARS-CoV-2. However, the RaTG13 Spike protein only poorly interacts with ACE2, the human receptor of SARS-CoV-2. In an interdisciplinary study just published in Nature Communications, members of CRC 1279 together with cooperation partners show that a single T403R amino acid change allows the Spike protein of RaTG13 to interact with the human ACE2 receptor and to mediate infection of human lung cells. Vice versa mutation of R403T reduced infectivity of SARS-CoV-2 S pseudotyped reporter virus and recombinant SARS-CoV-2 replication.
"We were surprised that a single mutation has such a strong impact infectivity and cell tropism of the RaTG13 Spike protein" explains Zech, first author of the study. Computational modeling and site-directed mutagenesis were utilized to determine the underlying molecular mechanism further. "A positively charged amino acid at position 403 allows the Spike protein to interact with the negatively charged E37 in human ACE2, therefore strengthening the interaction of virus glycoprotein and its cellular receptor".
While T403R enhanced the ability of the bat coronavirus Spike to bind to human cells, the mutant Spike protein of the bat coronavirus was still efficiently neutralized by sera from individuals vaccinated against COVID-19. Altogether, the study helps to better predict the zoonotic potential of animal coronaviruses and suggests that current vaccination regimens might protect against future viral zoonoses.
Zech et al. (https://pubmed.ncbi.nlm.nih.gov/34824253/)